A new era of financial disclosure and scrutiny for doctors has begun with all payments from pharma companies now displayed on a single publicly searchable website.
The disclosureaustralia.com.au site lists all payments and financial support provided by Medicines Australia member companies to physicians in Australia.
The site includes information – supplied by pharma companies – on payments such as speaker fees for educational meetings, payments for being on an advisory board, and financial support for attending medical conferences including airfares, accommodation and registration fees.
The information was previously available via individual company websites, but the new Centralised Reporting System presents it in a single place in an easily searchable form open to the public, according to Medicines Australia.
“We are taking our already rigorous transparency model to the next level by providing a platform that’s easy to use and accessible to all,” said CEO Elizabeth de Somer.
“Our members provide clear information about payments to a healthcare professional so they can access the latest educational opportunities, attend conferences or symposia, or for their services as an expert.”
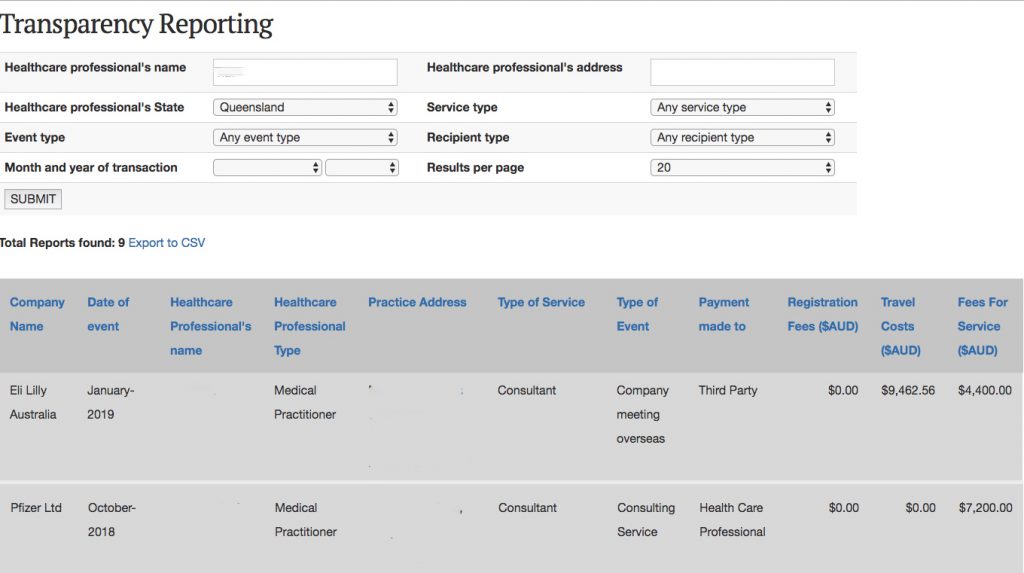 The website allows users to search for the name of an individual physician and view all the payments made to them reported by pharma companies. Details include the name of the company, the amount of money, the reason for the payment (speaker fee, conference attendance etc) and whether it was paid directly to the physician or to a third party. Updated information will be published in February and August each year.
The website allows users to search for the name of an individual physician and view all the payments made to them reported by pharma companies. Details include the name of the company, the amount of money, the reason for the payment (speaker fee, conference attendance etc) and whether it was paid directly to the physician or to a third party. Updated information will be published in February and August each year.