Pacemakers and cochlear implants seemed like the stuff of science fiction when they were first introduced, and the newest generation of medical implants are just as mind-boggling
A century ago, placing man-made parts into a human body to replicate lost function or prevent debilitating symptoms would have been viewed as science fiction.
Today implantable electronic medical devices (IEMDs), like pacemakers and cochlear implants, are widely accepted. But the next generation of these devices, powered by artificial intelligence, will once again push the limits of our imagination.
THE FIRST PATIENT
In 1958, 43-year old Arne Larsson of Sweden became the world’s first implantable pacemaker patient after a viral infection damaged his heart.
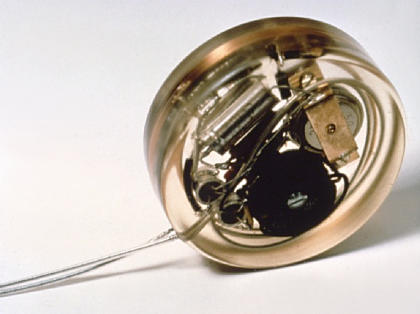
Dr. Åke Senning, a heart surgeon and Dr. Rune Elmqvist, an engineer at Elema-Schönander electronics company (later Siemens-Elema), rigged an experimental device that generated electrical pulses to prompt his heart to beat at a normal rate.
Shortly thereafter, in 1960, American engineer Wilson Greatbatch and his coworkers successfully implanted pacemakers in 10 patients.
Arne Larsson’s cardiac implant was the start of a whole new field of medicine. Today millions of procedures are performed around the world each year to place implantable electronic medical devices into patients. Some, such as pacemakers, are life-saving, while others, like the cochlear implant, significantly improve a patient’s quality of life.
Nearly 70 years later, most contemporary IEMDs still feature very similar technology to those early cardiac implants, and are in general hermetically sealed electrical stimulation and/or recording devices that interface with the relevant body tissue via electrodes (metal alloy contacts).
As microprocessors become more powerful and energy efficient, implant capabilities have improved. Modern generations of cardiac IEMDs, for example, act as both a pacemaker and a defibrillator (monitoring the heart for abnormal electrical patterns and responsively defibrillating to prevent heart attacks).
THE LEGACY OF THE COCHLEAR IMPLANT
In 1978, Professor Graeme Clark and colleagues at the University of Melbourne implanted the first cochlear implant, following more than a decade of research.
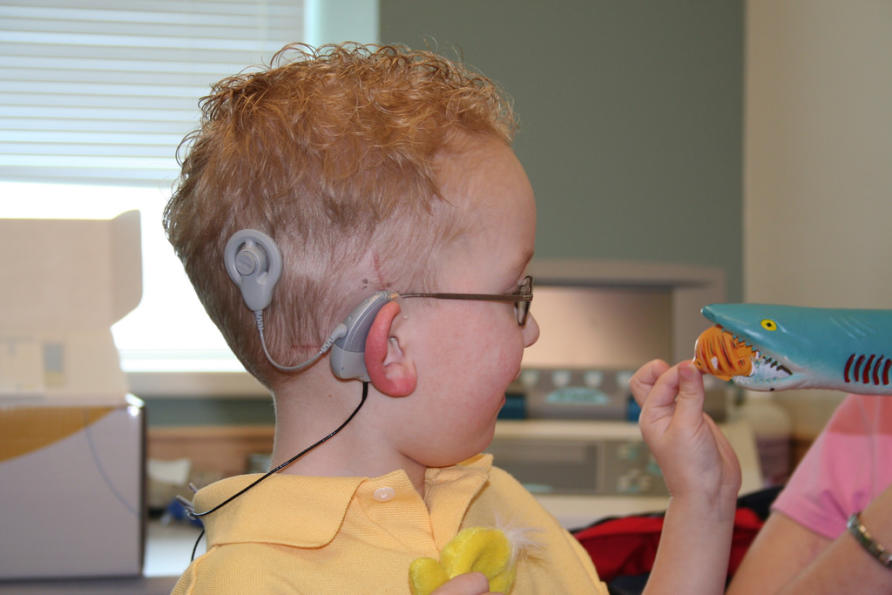
In retrospect, the cochlear implant was the first generation of ‘smart’ IEMDs used clinically. The researchers developed a way to convert digitally coded sound into electrical stimulation of the auditory nerve, to artificially replicate sound perception.
The ‘smart’ external unit, fitted to the patient’s outer ear, consists of microphones to capture sounds and a processor to convert the sounds into digital information. The information is transferred through the induction coil to the implant to stimulate the auditory nerve.
This triumph in bionics research created a legacy among researchers that is still very much alive today, with the University of Melbourne’s Graeme Clark Institute of Biomedical Engineering leading research on new devices that, just like pacemakers in the 50s, seem like the stuff of science fiction right now.
IMPLANTABLES FOR THE 21ST CENTURY
There are a whole range of IEMDs currently in use, such as phrenic nerve stimulation to restore breathing function in patients with breathing disorders, glucose sensors for diabetics, sacral nerve stimulation for patients with bladder disorders, and implantable drug delivery systems.
