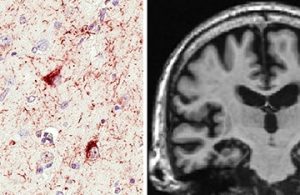
Limbic-predominant Age-related TDP-43 Encephalopathy, or LATE, as seen by microscope and MRI. Images courtesy of Nelson et. al. and Brain.
The first diagnostic criteria and guidance have been developed for a newly recognised form of non-Alzheimer dementia known as limbic-predominant age-related TDP-43 encephalopathy (LATE).
Published in Brain, the consensus-based recommendations on LATE describe it as a dementia syndrome typically affecting older patients with neuropathological change defined by a stereotypical TDP-43 proteinopathy, with or without coexisting hippocampal sclerosis pathology.
An international expert consensus group, led by Professor Peter Nelson of the University of Kentucky, say that emerging research seems to indicate that TDP-43 (transactive response DNA binding protein of 43 kDa) is associated with a high proportion of non-Alzheimer dementia.
The article notes that about 25% of brains from autopsy cohorts of older adults have sufficient burden of LATE pathology to be associated with discernible cognitive impairment.
Previous research has shown that TDP-43 has a causative role in most cases of amyotrophic lateral sclerosis and frontotemporal lobar degeneration.
“LATE is distinguished from frontotemporal lobar degeneration with TDP-43 pathology based on its epidemiology (LATE generally affects older subjects), and relatively restricted neuroanatomical distribution of TDP-43 proteinopathy,” says the report co-authored by Professor Glenda Halliday of the Brain and Mind Institute at Sydney University.
“Given that the ‘oldest-old’ are at greatest risk for LATE-NC, and subjects of advanced age constitute a rapidly growing demographic group in many countries, LATE has an expanding but under-recognised impact on public health,” the authors say.
The report says LATE-NC appears to affect the medial temporal lobe structures preferentially, and people with the condition also have atrophy in the medial temporal lobes, frontal cortex, and other brain regions.