Science reporter Maryanne Demasi was last month cleared of allegations of research misconduct in relation to her University of Adelaide PhD thesis published in 2004.
The allegations were investigated by an independent panel convened by the University of Adelaide’s Office of Research Ethics, Compliance and Integrity. It comprised a former justice of the Supreme Court of South Australia and three professors of medical science not linked with the University of Adelaide.
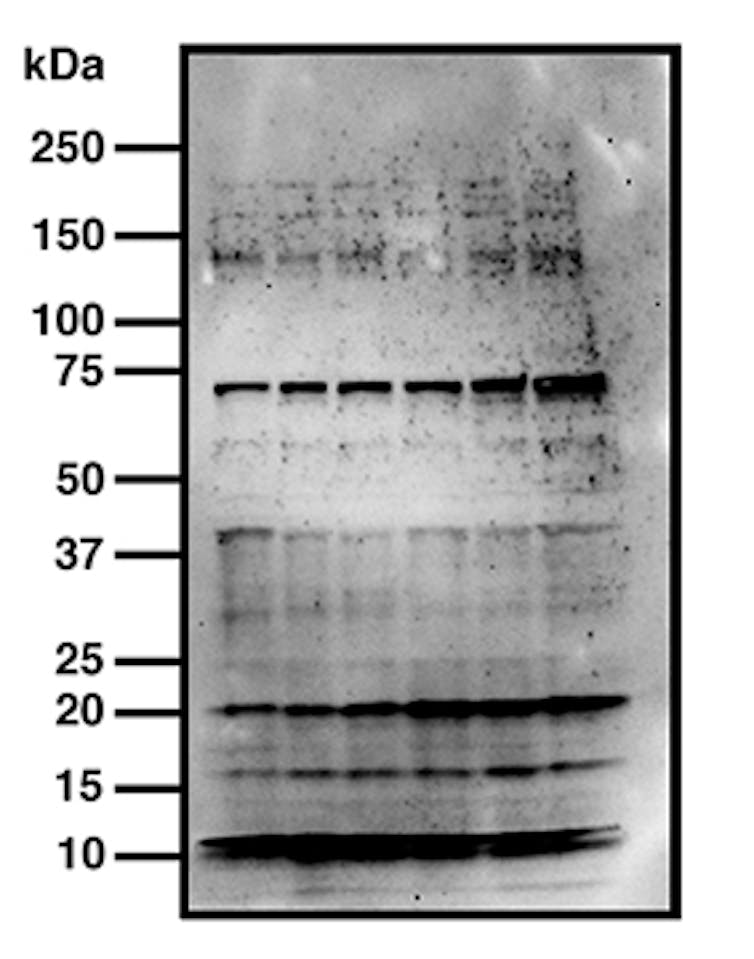
There were 17 allegations about images in Demasi’s thesis. The question before the panel was whether the images “had been duplicated and/or manipulated in a manner that deviated from the applicable standards at the relevant time”.
All of the impugned images depicted the results of experiments to detect proteins or RNA molecules. The techniques to do this are known as “western blot” and “northern blot”, respectively.
In 14 cases the panel found no image duplication had taken place. In the remaining three cases Demasi “admitted that she had duplicated or probably duplicated the relevant impugned images” – but the panel “could not be satisfied that the duplication constituted a deviation from the applicable standards at the relevant time”.
It’s not clear whether blot images are the focus of The Journal of Biological Chemistry’s expression of concern in 2017 regarding one of Demasi’s scientific papers published in 2003.
Demasi firmly denies any research misconduct in both her thesis and research papers. But what are western and northern blots? And why do they attract such scrutiny?
Detecting proteins
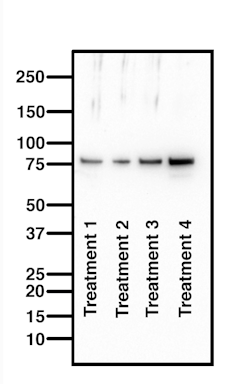
Westerns blots detect proteins, and are a commonly used and sensitive scientific technique. The image they produce may look like a nondescript smudge, but western blots are in fact a very useful way to home in on a specific protein in a complex mixture of biological materials, such as blood or tissue.
Western blots are used in the diagnosis of Lyme disease, for example. This is because infection with the bacterium that causes Lyme (Borrelia burgdorferi) leaves a unique fingerprint of proteins in a patient’s blood.
Western blots also enable researchers to determine changes in proteins in response to a drug regimen. In my own lab, I routinely run western blots to measure changes in protein profiles in response to a dietary supplement that we are trialling for the treatment of motor neuron disease.
Western, southern, northern
Western blots are named for the location of the American lab where they were first conducted. The choice of the term “western” is apparently in homage to the previously named Southern (named after Ed Southern) and northern blots, for detecting DNA and RNA, respectively.
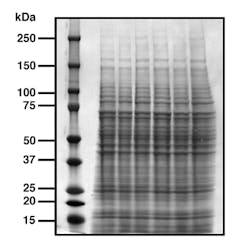
To run a western blot, a mix of proteins is loaded onto a gel made of polyacrylamide that allows separation of the proteins according to their molecular weights. Molecular weight is measured in kilodaltons, or kDa (the dalton or atomic mass unit is defined as 1/12 the weight of the carbon-12 isotope, or approximately one hydrogen atom).