
Dr Frank Lin
A precision oncology guide released this week will provide a pragmatic algorithm for therapeutic decision-making by Australian oncologists faced with a molecular report, its developers say.
The TOPOGRAPH (Therapy-Oriented Precision Oncology Guidelines for Recommending Anti-cancer Pharmaceuticals) database is an online compendium that incorporates the latest precision oncology research – such as druggable targets – to help clinicians recommend the best treatments in cancer medicine.
“It should assist oncologists with rapid identification of the relevant literature when encountering an unfamiliar biomarker, promoting structured communication with patients about potential therapies, and in facilitating the search for clinical trial linking a molecular biomarker to investigational therapies,” its developers say.
It also captures important Australian-specific ‘real-world’ aspects of decision-making pertaining to drug treatments such as local access, according to oncologists and researchers at the Garvan Institute of Medical Research and The Kinghorn Cancer Centre, Sydney.
“TOPOGRAPH is uniquely useful in the Australian context because it combines up-to-date information on treatments approved for use in Australia in both clinical and trial settings,” said Dr Frank Lin, Garvan Senior Research Officer who led the development of the platform.
Dr Lin said there were already several precision oncology resources available that interpret the potential therapeutic significance of genomic variations and other biomarkers in cancer. However they did not account for real world factors such as government subsidies and approvals by national regulators, which could limit access to treatments.
“We designed this tool because there’s no good alternatives in Australia to help oncologists sieve through potential treatment options when facing a complex genomic report,” he said.
Oncologists could look up any of the parameters on the platform, he said, as well as combinations of biomarkers, cancer types, and therapies to view information tailored for patients with advanced cancer.
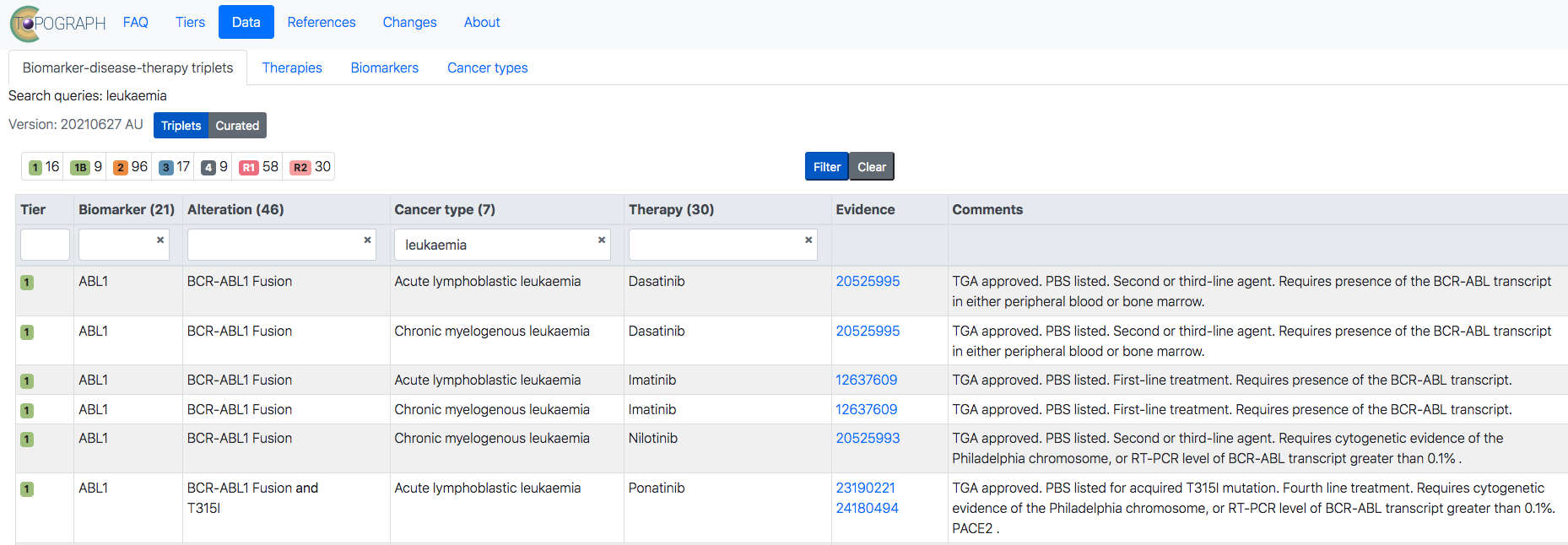 Published in npj Precision Oncology, the TOPOGRAPH compendium includes 373 therapies, 199 biomarkers, and 106 cancer types.
Published in npj Precision Oncology, the TOPOGRAPH compendium includes 373 therapies, 199 biomarkers, and 106 cancer types.
The TOPOGRAPH team, which also included researchers from St Vincent’s Hospital, Sydney, the Australian Genomic Cancer Medicine Program (Omico), UNSW Sydney, Sydney University and the NHMRC Clinical Trials Centre, categorised each biomarker-disease-therapy triplet using a tiering system reflective of key therapeutic considerations:
- Approved and reimbursed therapies with respect to a jurisdiction (Tier 1),
- Evidence of efficacy or approval in another jurisdiction (Tier 2),
- Evidence of antitumour activity (Tier 3), and
- Plausible biological rationale (Tier 4).
Two resistance categories were defined: lack of efficacy (Tier R1) or antitumor activity (Tier R2).
Of the therapies catalogued in TOPOGRAPH, 24% and 19% are designated Tiers 1 and 2, respectively, while 79% of therapies are supported by preclinical studies, early clinical trials, retrospective studies, or case series (Tiers 3 and 4).
Dr Subo Thavaneswaran, medical oncologist at The Kinghorn Cancer Centre and Garvan researcher, said the database was already proving useful in a clinical setting.
“Applying TOPOGRAPH to our Molecular Tumour Board recommendations at The Kinghorn Cancer Centre gives us greater confidence in their consistency, evidence-base and understanding of accessibility to therapies in the Australian context,” he said
The researchers plan to update TOPOGRAPH with treatments and therapies as they emerge and undergo assessment by the TGA.
It also has the potential to support translational research, by informing the design of new correlative studies to explore more precise biomarkers for targeted therapies, they suggested.